5.1 Weathering
Mechanical Weathering
Intrusive igneous rocks form at depths of several hundreds of metres to several tens of kilometres. Sediments are turned into sedimentary rocks only when they are buried by other sediments to depths in excess of several hundreds of metres. Most metamorphic rocks are formed at depths of kilometres to tens of kilometres. Weathering cannot even begin until these rocks are uplifted through various processes of mountain building—most of which are related to plate tectonics—and the overlying material has been eroded away and the rock is exposed as an outcrop.
The most important agents of mechanical weathering are:
- The decrease in pressure that results from removal of overlying rock
- Freezing and thawing of water in cracks in the rock
- Cracking from plant roots and removal of material by burrowing animals
- Formation of salt crystals within the rock
When a mass of rock is exposed by weathering and removal of the overlying rock, there is a decrease in the confining pressure on the rock, and the rock expands. This unloading promotes cracking of the rock, known as exfoliation.
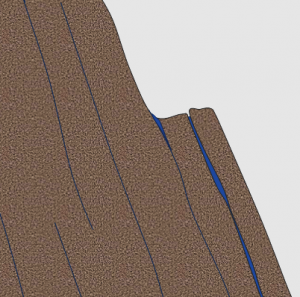
Frost wedging is the process by which water seeps into cracks in a rock, expands on freezing, and thus enlarges the cracks (Figure 5.1.1). The effectiveness of frost wedging is related to the frequency of freezing and thawing. Frost wedging is most effective in a climate like Canada’s. In warm areas where freezing is infrequent, in very cold areas where thawing is infrequent, or in very dry areas, where there is little water to seep into cracks, the role of frost wedging is limited. If you are ever hiking in the mountains you might hear the effects of frost wedging when the Sun warms a steep rocky slope and the fragments of rock that were pried away from the surface by freezing the night before are released as that ice melts.
In many parts of Canada, the transition between freezing nighttime temperatures and thawing daytime temperatures is frequent — tens to hundreds of times a year. A common feature in areas of effective frost wedging is a talus slope—a fan-shaped deposit of fragments removed by frost wedging from the steep rocky slopes above (Figure 5.1.2).

A related process, frost heaving, takes place within unconsolidated materials on gentle slopes. In this case, water in the soil freezes and expands, pushing the overlying material up. Frost heaving is responsible for winter damage to roads all over North America.
Mechanical weathering is greatly facilitated by erosion, which is the removal of weathering products, allowing for the exposure of more rock for weathering. A good example of this is shown in Figure 5.1.2. On the steep rock faces at the top of the cliff, rock fragments have been broken off by ice wedging, and then removed by gravity. This is a form of mass wasting. Other important agents of erosion that also have the effect of removing the products of weathering include water in streams (Lab 8), glacial ice, and waves on the coasts.
Chemical Weathering
Chemical weathering results from chemical changes to minerals that become unstable when they are exposed to surface conditions. The kinds of changes that take place are highly specific to the mineral and the environmental conditions. Some minerals, like quartz, are virtually unaffected by chemical weathering, while others, like feldspar, are easily altered. In general, the degree of chemical weathering is greatest in warm and wet climates, and least in cold and dry climates. The important characteristics of surface conditions that lead to chemical weathering are the presence of water (in the air and on the ground surface), the abundance of oxygen, and the presence of carbon dioxide, which produces weak carbonic acid (H2CO3) when combined with water. That process, which is fundamental to most chemical weathering, can be shown as follows:
|
H2O + CO2 ↔ H2CO3 then H2CO3 ↔ H+ + HCO3− water + carbon dioxide ↔ carbonic acid then carbonic acid ↔ dissolved hydrogen ions + dissolved bicarbonate ions |
Yikes! Chemical formulas
Lots of people seize up when they are asked to read chemical or mathematical formulas. It’s OK, you don’t necessarily have to! If you don’t like the formulas just read the text underneath them. In time you may get used to reading the formulas.
The double-ended arrow “↔ ” indicates that the reaction can go either way, but for our purposes these reactions are going towards the right.
Here we have water (e.g., as rain) plus carbon dioxide in the atmosphere, combining to create carbonic acid. Then carbonic acid dissociates (comes apart) to form hydrogen and bicarbonate ions. The amount of CO2 in the air is enough to make weak carbonic acid. There is typically much more CO2 in the soil, so water that percolates through the soil can become more acidic. In either case, this acidic water is a critical to chemical weathering.
In some types of chemical weathering the original mineral becomes altered to a different mineral. For example, feldspar is altered—by hydrolysis—to form clay minerals plus some ions in solution. In other cases the minerals dissolve completely, and their components go into solution. For example, calcite (CaCO3) is soluble in acidic solutions.
The hydrolysis of feldspar can be written like this:
|
CaAl2Si2O8 + H2CO3 + ½O2 ↔ Al2Si2O5(OH)4 + Ca2+ + CO32− plagioclase feldspar + carbonic acid ↔ kaolinite + dissolved calcium ions + dissolved carbonate ions |
This reaction shows calcium-bearing plagioclase feldspar, but similar reactions could also be written for sodium or potassium feldspars. In this case, we end up with the mineral kaolinite, along with calcium and carbonate ions in solution. Those ions can eventually combine (probably in the ocean) to form the mineral calcite. The hydrolysis of feldspar to clay is illustrated in Figure 5.1.3, which shows two images of the same granitic rock, a recently broken fresh surface on the left and a clay-altered weathered surface on the right. Other silicate minerals can also go through hydrolysis, although the end results will be a little different. For example, pyroxene can be converted to the minerals chlorite or smectite, and olivine can be converted to the mineral serpentine.

Oxidation is another very important chemical weathering process. The oxidation of the iron in a ferromagnesian silicate starts with the dissolution of the iron. For olivine, the process looks like this, where olivine in the presence of carbonic acid is converted to dissolved iron, carbonate, and silicic acid:
|
Fe2SiO4+ 4H2CO3 ↔ 2Fe2+ + 4HCO3− + H4SiO4 olivine + (carbonic acid) ↔ dissolved iron ions + dissolved carbonate ions + dissolved silicic acid |
But in the presence of oxygen and carbonic acid, the dissolved iron is then quickly converted to the mineral hematite:
|
2Fe2+ + 4HCO3− + ½ O2 + 2H2O ↔ Fe2O3 + 4H2CO3 dissolved iron ions + dissolved bicarbonate ions + oxygen + water ↔ hematite + carbonic acid |
The equation shown here is for olivine, but it could apply to almost any other ferromagnesian silicate, including pyroxene, amphibole, or biotite. Iron in the sulphide minerals (e.g., pyrite) can also be oxidized in this way. And the mineral hematite is not the only possible end result, as there is a wide range of iron oxide minerals that can form in this way. The results of this process are illustrated in Figure 5.1.4, which shows a granitic rock in which some of the biotite and amphibole have been altered to form the iron oxide mineral limonite.

A special type of oxidation takes place in areas where the rocks have elevated levels of sulphide minerals, especially pyrite (FeS2). Pyrite reacts with water and oxygen to form sulphuric acid, as follows:
|
2FeS2 + 7O2 + 2H2O ↔ 2Fe2+ H2SO4 + 2H+ pyrite + oxygen + water ↔ dissloved iron ions + sulphuric acid + dissolved hydrogen ions |
The runoff from areas where this process is taking place is known as acid rock drainage (ARD), and even a rock with 1% or 2% pyrite can produce significant ARD. While ARD does occur naturally when rocks containing pyrite are exposed at the Earth’s surface, some of the worst examples of ARD are at mine sites. These sites can have significant problems with ARD, especially where pyrite-bearing rock and waste material have been mined from deep underground and then piled up and left exposed to water and oxygen. One example of that is the Mt. Washington Mine on Vancouver Island in B.C. (Figure 5.1.5), but there are many similar sites across Canada and around the world.

The hydrolysis of feldspar and other silicate minerals and the oxidation of iron in ferromagnesian silicates all serve to create rocks that are softer and weaker than they were to begin with, and thus more susceptible to mechanical weathering.
The weathering reactions that we’ve discussed so far involved the transformation of one mineral to another mineral (e.g., feldspar to clay), and the release of some ions in solution (e.g., Ca2+ or Fe2+). Some weathering processes involve the complete dissolution of a mineral. Calcite, for example, will dissolve in weak acid, to produce calcium and bicarbonate ions. The equation is as follows:
|
CaCO3 + H+ + HCO3− ↔ Ca2+ + 2HCO3− calcite + dissolved hydrogen ions + dissolved bicarbonate ions ↔ dissolved calcium ions + dissolved bicarbonate ions |
Calcite is the major component of limestone (typically more than 95%), and under surface conditions, limestone can dissolve completely. Limestone also dissolves at relatively shallow depths underground, forming limestone caves.
Practice Exercise 5.1 Chemical Weathering
The main processes of chemical weathering are hydrolysis, oxidation, and dissolution. Indicate which process is primarily involved during each of the following chemical weathering changes:
- Pyrite to hematite
- Calcite to calcium and bicarbonate ions
See Appendix 2 for Practice Exercise 5.1 answers.
Media Attributions
- Figures 5.1.1, 5.1.3, 5.1.4, 5.1.5: © Steven Earle. CC BY.
- Figure 5.1.2: © Siobhan McGoldrick. CC BY.
To a geologist, an outcrop is an exposure of bedrock, the solid rock of the crust.
the physical breakdown of rock into smaller fragments
the situation where the expansion of freezing water pries rock apart
a sloped deposit of angular rock fragments at the base of a rocky escarpment or cliff
the action of surface processes (like running water or wind) that remove soil, rock, or dissolved material from one location on or near the Earth's surface, and then transports it to another location.
the mass failure, by gravity, of rock or unconsolidated material on a slope
a reaction between a mineral and water in which H+ ions are added to the mineral and a chemically equivalent amount of cations are released into solution
a hydrous sheet silicate mineral that typically exists as clay-sized grains
the reaction between a mineral and oxygen
Referring to a silicate mineral that contains iron and or magnesium.
the production of acid from oxidation of sulphide minerals (especially pyrite) in either naturally or anthropogenically exposed rock

