Hill-Langmuir equation
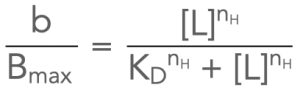 This is an equation for a hyperbola that allows you to determine the proportion of total receptors (or other proteins) bound at any given concentration of a ligand, [L], if you know the KD for the ligand interacting with those receptors. In order for binding to show hyperbolic behaviour, the KD for the interaction between ligand and receptor is typically much higher than the concentration of receptors present. Since a therapeutic effect is typically only evident when drug is interacting with a reasonable proportion of the total receptor population, the result of this is that concentrations of drug present are almost always far higher than the concentrations of receptor targets present, and so when a small amount of the free drug binds to the receptors, there is no appreciable change in the concentration of free (unbound) drug.
This is an equation for a hyperbola that allows you to determine the proportion of total receptors (or other proteins) bound at any given concentration of a ligand, [L], if you know the KD for the ligand interacting with those receptors. In order for binding to show hyperbolic behaviour, the KD for the interaction between ligand and receptor is typically much higher than the concentration of receptors present. Since a therapeutic effect is typically only evident when drug is interacting with a reasonable proportion of the total receptor population, the result of this is that concentrations of drug present are almost always far higher than the concentrations of receptor targets present, and so when a small amount of the free drug binds to the receptors, there is no appreciable change in the concentration of free (unbound) drug.
The equation shown contains an exponential term, nH. This is the Hill coefficient, and for most drugs, it can be assumed to equal 1 and can thus be ignored. For a few ligands that bind to dimeric or multimeric proteins with multiple subunits and multiple ligand binding sites, binding of one ligand to the protein complex may impact binding of further ligands to further binding sites within the same protein complex, either enhancing or attenuating binding. This phenomenon, called cooperativity of binding, is also seen physiologically, for example in the binding of oxygen to haemoglobin. Cooperativity yields binding curves and dose-response curves that are not simple hyperbolas. The figures below show binding curves on linear (left) and logarithmic (right) axes for three ligands that share a common KD value, but with differing Hill coefficients.
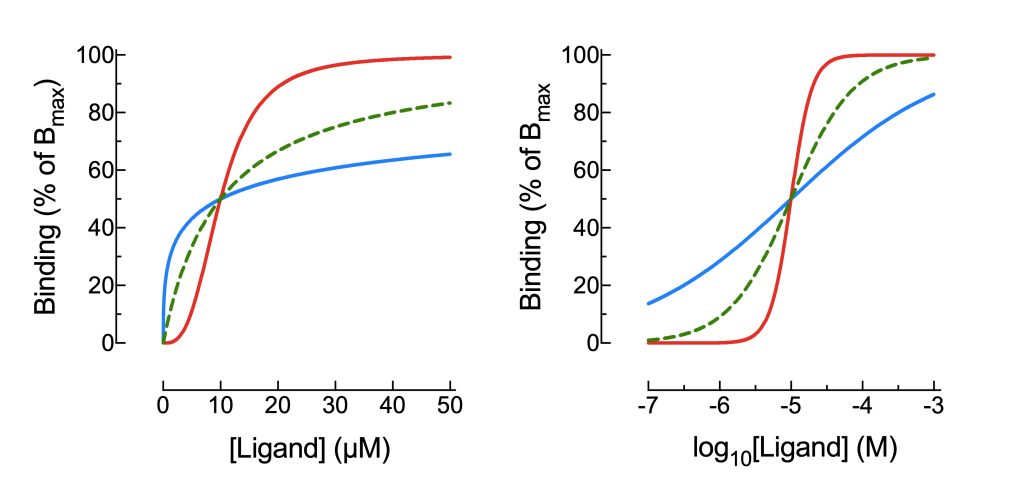
From a clinical therapeutic perspective, the target range for the steady state concentration of a cooperative drug may be narrower, or wider, than for most drugs, but this would be reflected in the recommended dosing regimens, and cooperativity is not an issue you need to be concerned with in relation to clinical use of drugs.
In those situations where a ligand has extremely high affinity for a receptor (such that the KD is down in the same range as the concentration of receptors) OR when the concentration of protein “targets” is extremely high (such as with drug binding to plasma proteins) then depletion of free ligand becomes significant (this is why plasma protein binding has such a significant impact on drug behaviour), and binding is described by a quadratic equation. It is not necessary to be familiar with this equation, but you should be aware that drugs with extremely high affinities for their targets do not follow the same mathematical patterns of pharmacodynamic behaviour seen with most drugs.

